How much do we know?
Let’s be clear at the outset: Medical experts and scientists agree that we still don’t have enough evidence that marijuana use can directly cause psychosis. Recent studies suggest a close relationship in people at risk, but more evidence is needed .
Thus, people with a family history of psychosis/schizophrenia or other vulnerability are advised to avoid marijuana. Strong-acting strains and smoking marijuana at a younger age are considered particularly risky. However, most studies have only dealt with associations, which means a causal relationship has not been established. For example, just because psychosis was associated with marijuana use does not mean that marijuana causes psychosis.
Also, even if research has shown that marijuana use contributes to psychosis, it is unlikely that this is the only cause. Complex disorders such as schizophrenia are always related to many possible factors, including genetics, brain chemistry, environment, and health conditions, which can vary from one person to another.
Common factors
Research shows that many factors can alter the relationship between marijuana and psychosis, including:
-
Types of marijuana varieties and their effectiveness
-
Patterns of use
-
Age of use
-
Genetics
-
Childhood trauma
-
Lifestyle
-
Gender
-
Race
-
Social and cultural influences
-
Other illnesses and/or addictions
Adverse effects of cannabis
The most common side effects caused by smoking cannabis or using a mouth spray containing cannabis extract are:
-
Headache
-
Dizziness
-
Drowsiness
-
Fatigue
-
Dry mouth
-
Nausea
-
Paranoid thinking and dissociation
A number of unconfirmed reports suggest that cannabis can cause a range of acute psychotic symptoms that include depersonalization, derealization, paranoia, anxiety, flight of ideas, disorganized thinking, delusions of persecution, grandiose delusions, auditory and visual hallucinations, attention disorders and memory.
Cannabis-induced psychosis

Small studies on healthy volunteers show that THC (from cannabis) can cause psychosis-like symptoms. However, some people seem to be more vulnerable to these negative effects than others. According to large recent studies in Europe and Brazil, daily smoking of potent marijuana was associated with an increased risk of developing psychosis almost five times that of people who had never used marijuana.
Another study involving 6,700 people concluded that people with marijuana-induced psychosis had a 47 percent chance of developing bipolar disorder or schizophrenia within three to five years. Substance-induced psychosis at a young age was associated with a higher risk of conversion to schizophrenia. Self-harm following substance-induced psychosis was associated with a higher risk of both schizophrenia and bipolar disorder.
On the other hand, no one knows for sure if cannabis causes schizophrenia (or bipolar disorder). Scientists say that people who are predisposed to schizophrenia may also be more likely to use cannabis. They are also more likely to smoke cigarettes.
However, another theory claims that some common factors may underlie both cannabis use and psychosis. Finally, a group of researchers believe that cannabis use in people with mental health problems is a way to “self-medicate” and alleviate the early symptoms of schizophrenia.
Effect of THC on neurotransmitters
Below are experimental and theoretical data related to the effects of THC on the neurotransmitter system.
Dopamine
Animal experiments show that THC increases dopamine in the striatum, which is thought to be a biological feature of schizophrenia and psychosis. Researchers believe that dopamine in the striatum can increase motivation. But if dopamine levels in the striatum are already high, cannabis can push it to its limit.
Activation of the CB1 receptor can also increase dopamine in the prefrontal cortex, which can lead to its nonspecific activation and disruption of normal signal processing, leading to poor cognitive integration of input. Scientists say that excessively high levels of dopamine may contribute to working memory deficits associated with cannabis exposure.
Structural and functional imaging studies show that chronic cannabis use causes the same negative changes in brain structure as in schizophrenia, such as decreased hippocampal and amygdala volume. According to one experimental hypothesis, this may be due to the neurotoxic effects of chronically elevated dopamine levels.
GABA
Activation of the CB1 receptor reduces the release of GABA in hippocampal neurons in animals. This is thought to disrupt the synchronization of neuronal activity, which interferes with memory consolidation and the brain’s ability to make normal associations, ultimately leading to psychotic symptoms.
In other words, some scientists claim that GABA can stop “crazy” thoughts, but in some key places it is reduced when CB1 receptors are activated. Limited research suggests that people with schizophrenia may have lower levels of GABA in the brain, but more research is needed.
Glutamate
According to one theory, so-called cognitive gateway functions in the brain prevent illogical thoughts from entering the mind.
Animal studies show that cannabinoids reduce glutamate levels (and NMDA activation) in several brain regions involved in the regulation of gateway functions, such as the hippocampus, prefrontal cortex, adjoining nucleus and amygdala. Ketamine, an NMDA blocker, can also induce psychosis, highlighting the potential role of lower glutamate levels in certain areas and psychosis.
Genes and cannabis-induced psychosis

The following SNPs were found to be associated only with cannabis-induced psychosis. This does not mean that their presence increases the risk of cannabis-induced psychosis. More work is needed before the effect of cannabis use on psychosis in people with different genetic variations can be elucidated.
DRD2 (dopamine gene)
Limited experimental evidence suggests that dopamine D2 receptors and schizophrenia are closely related. There is an association between psychosis and relatively large numbers of D2 receptors in the striatum, a dopamine-rich region of the brain. In addition, DRD2 receptors are blocked by neuroleptics, which help control the symptoms of psychosis.
Scientists say that relatively excessive activation of dopamine D2 can lead to decreased “gateway” function. It is that people have reduced filtering of relevant information (reduced signal-to-noise ratio) as well as blocked distractions. These brain processes contribute to various higher-order cognitive functions and are actively involved in attention control.
Some studies show that patients with schizophrenia who perform attention tasks have less activity and lower levels of gray matter in the cingulate gyrus, an area of the brain that strongly influences attention processing and executive function. This area is influenced by D2 receptors.
SNP
The “A” allele of rs1076560 in the dopamine DRD2 gene was associated with cannabis-induced psychosis in one study. In another study, it was associated with a 10-fold increased risk of psychosis in daily cannabis users. This SNP has also been linked to behavior and brain activity during cognitive and emotional processing in healthy subjects and in patients with schizophrenia.
In healthy subjects, the interaction between the A DRD2 rs1076560 allele and the A AKT1 rs1130233 allele was associated with decreased AKT1 protein levels and increased GSK-3β, as well as an altered cingulate response and impaired cognitive function during attention processing.
GSK3b is a protein that is thought to increase oxidative stress and inflammation in the brain. Certain factors may inhibit this. D2 stimulation by dopamine inhibits AKT1 , which can increase GSK3b and has been associated with psychosis in limited studies.
AKT1 levels in the prefrontal cortex in patients with schizophrenia have been reduced in some brain imaging studies. It has been suggested that antipsychotics help by increasing AKT1 and decreasing GSK-3β, but this has not been proven.
CB1 receptor (cannabinoid gene)
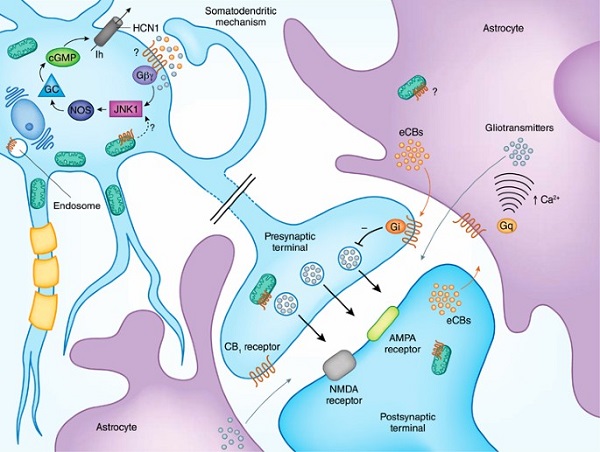
The cannabinoid receptor type 1 (CB1) is the most well characterized cannabinoid receptor. Scientists are investigating whether changes in this receptor may increase the risk of schizophrenia. The cannabinoid system plays an important role in brain maturation during adolescence, which is a critical period for the maturation of many neurotransmitters, including dopamine.
Some scientists suggest that abnormal CB1 function may interfere with the maturation of neural networks during adolescence, which may contribute to the later development of psychosis. However, this has yet to be tested in large samples of people. There have been conflicting data on schizophrenia. For example, some studies have reported increased cannabinoid receptor (CB1) function in the dorsolateral prefrontal cortex and cingulate gyrus, which is associated with volume loss in these areas. The CB1 receptor SNP rs12720071 (allele “C”) has been associated with lower white matter volumes (frontal and temporal) and an increased risk of schizophrenia during marijuana abuse in other studies.
Conclusion
Researchers in the field need to carefully balance research on cannabis and schizophrenia between the neurophysiology and social aspects of cannabis-induced psychosis. Since cannabis use and the development of psychosis depend on both social and biological factors, it is important that major institutions continue to conduct balanced research in this area.
The dose and frequency of cannabis use continue to be reliable indicators of the risk of developing psychosis. However, some people appear to be sensitive to low levels of cannabis exposure; it is still unclear whether this is due to increased cannabis potency or some other factor, such as genetics.
Overall, we still do not have enough information and knowledge about who is at risk of developing cannabis psychosis prior to cannabis exposure to reliably develop a prevention strategy for such syndromes. As more and more U.S. states allow access to cannabis for recreational or medical purposes, this represents a great naturalistic experiment with these populations, and it will be several years before we can judge the impact of these regulatory changes on health, morbidity, and prevalence of psychosis.