THC use can affect a person’s fertility depending on gender, general health, socioeconomic status, and several other factors. Here we will review existing research on the effects of THC on human fertility in order to provide readers with an accurate and up-to-date summary of the current state of scientific knowledge.
To understand how THC can affect fertility, we must understand its effects specifically on men and women. We will begin by examining the effects on the male reproductive system, and then focus on the more complex effects on female fertility.
THC and male fertility
Some studies of the effects of THC use on male fertility have shown that regular cannabis use can reduce spermatogenesis (sperm production in the testicles) and testosterone levels.
In 2012, the American Society of Andrology published a review of studies on the effects of illicit drug use on male fertility. The researchers found that most studies consistently concluded that cannabis use had a negative impact on male reproductive physiology.
Another study by Vescovi et al. in 1992 showed that levels of luteinizing hormone (LH), an important pituitary hormone involved in reproductive function, were reduced in male chronic cannabis users compared to the control group (the control group did not use cannabis). The results of a previous study conducted in 1986 also showed a significant decrease in LH levels immediately after smoking cannabis.
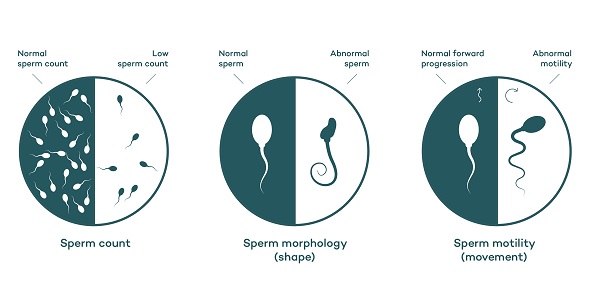
And an even earlier study of testosterone levels in “chronic” cannabis users showed that 6 of the 17 subjects had oligospermia (low sperm count) and the average testosterone level in the cannabis using group was just over half that of the control group (Kolodny et al, 1974). It was also observed in this study that the effect of cannabis on testosterone levels was dose dependent.
Male fertility and ECS

Apparently the endocannabinoid system plays an important role in regulating male reproductive processes: sperm count, testosterone levels and levels of other key hormones such as LH. For healthy adult men, THC use does cause some negative effects on fertility, which tend to increase with higher doses. However, the endogenous THC analogue, anandamide, appears to be crucial to the functioning of the male reproductive system.
Cannabinoid type I receptors were first found to be present in human testicles in a 1993 study .
In a 1993 study, CB1 receptors were first found to be present in human testicles, and later, in 2002, anandamide was found in human seminal fluid and cannabinoid type 1 receptors in human spermatozoa (found to bind the agonist CP-55,940 to spermatozoa).
The researchers concluded that the presence of THC and the synthetic analog of anandamide, AM-356, reduced sperm motility in vitro. Interestingly, AM-356 was found to have a biphasic dose-dependent effect on sperm motility, causing inhibition at high doses and hyperactivity at low doses.
Anandamide and “capacitation” of sperm
In 1994, scientists conducted a study in which it was discovered that human sperm cells are not actually capable of fertilizing oocytes (eggs) immediately after leaving the testicles. The culmination of this process requires a period of exposure to certain important hormones, enzymes and proteins on its way through the vas deferens and ejaculatory ducts of the male reproductive system (as well as the reproductive fluids of the female vagina and oviduct) before it becomes “capable” and can fertilize an egg.
Based on the above, scientists began to study the effects of various compounds on fertilization processes. In 2002, they provided convincing evidence that the presence of anandamide in seminal fluid and its ability to bind to CB1 receptors of sperm cells is the key to the “capacitation” of sperm cells on their way to the ejaculatory ducts. However, it must be present in certain “average” concentrations. If anandamide levels are too high, it can instead have a drastic inhibitory effect on the ability of spermatozoa to fertilize oocytes.
So anandamide is useful, but THC is not?
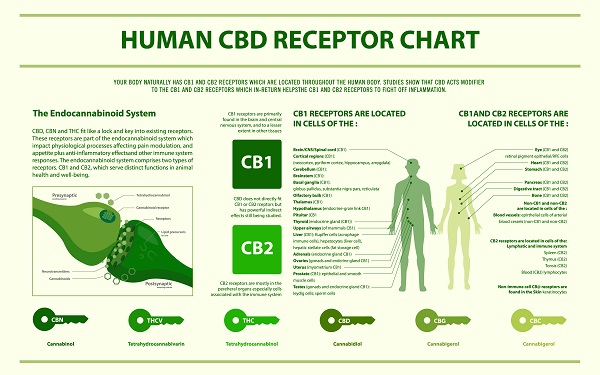
Although THC and anandamide are both CB1-receptor agonists, they are very different in structure and therefore affect certain metabolic processes differently.
Anandamide has a much shorter half-life than THC (only a few minutes for anandamide compared to 24 hours for THC). Thus, while anandamide degrades soon after contact with the receptor, THC can remain in nearby adipose tissues for a much longer time and can continue to stimulate receptors, ultimately causing overstimulation and potential negative effects.
As is often the case in cannabinoid science, the dosage decides everything, and it may be that very small doses of THC can benefit men who have reproductive problems that may be related to low anandamide levels.
THC and female fertility
While the effects of THC on male fertility seem pretty obvious – men who chronically use cannabis may experience some abnormalities in their reproductive physiology, the effects on the reproductive system of women are less obvious. Let’s break this down in detail.
Female reproductive health itself is extremely complex in terms of physiology, as it includes not only the ability to become pregnant, but also the ability to bear healthy offspring and to give birth successfully.
In studies that were just starting out, scientists found that THC use can disrupt the menstrual cycle, decrease oogenesis (production of eggs in the ovaries), and significantly reduce implantation and embryo development. Chronic and prolonged use of cannabis by women has repeatedly been associated with lower fetal weight (risk increased by 50%) and early spontaneous abortion.
However, the findings are not conclusive and require further analysis of the results. Either very few participants were included in the study or there were distorting factors (smoking tobacco, etc.). Interestingly, in this study, the scientists who studied the intrauterine effects of cannabis on the fetus concluded that there were no adverse risks to the fetus from THC use.
For example, Qazi’s 1985 article on fetal abnormalities following intrauterine exposure to THC discussed five infants whose mothers admitted to using cannabis before and during pregnancy and who were born with various symptoms of growth retardation, neurological dysfunction, and deformity. Although this provides us with useful information, such a small sample size is far from sufficient to draw specific conclusions, and correlation does not suggest causation.
A study by Geber & Schramm in the 1970s proved that THC use causes various fetal abnormalities. In that study, rabbits, rats, and hamsters were given large doses of crude cannabis extract (in doses up to 600 mg/kg once). Of course, humans never consume such doses, so from a comparative analytical point of view, the results of such a study are of no practical value.
In fact, many earlier studies pointing to a correlation between THC use are contradicted by the results of studies conducted in the last 5-10 years. It has been suggested that socioeconomic status and poverty levels are a more significant cause of low birth weight and poor development than the use of these cannabis. Certainly this does not mean that THC use does not have adverse effects on the fetus, but it lends weight to the view that the risks have been greatly overestimated and exaggerated because of anti-drug campaign policies.
ECS and female fertility
As in men, it is obvious that the endocannabinoid system plays an important role in female fertility.
-
Studies have proven that CB1 receptors are present in the ovaries, the uterine endometrium and the developing embryo.
-
Anandamide is present in the oviductal and follicular fluid (fluids found in the fallopian tubes and the follicular antral region; the follicular antral region is the chamber in which ovarian follicles produce oocytes or immature oocytes)
-
CB1- and CB2-receptors have also been found at all stages of oocyte development itself.
As mentioned earlier, mammalian sperm require a period of exposure to both male and female reproductive fluids to become “capacitated” and able to fertilize an egg. Thus, the presence of anandamide in female reproductive fluids clearly promotes sperm capacitation on their way to the fallopian tubes.
Anandamide and CB1 receptors not only help the sperm to move and ease its way to the fallopian tubes, where fertilization takes place, but also play a crucial role in ovulation and implantation of the fertilized gamete into the uterine mucosa.
In particular, it has been shown that high levels of anandamide are necessary for successful ovulation and lower levels are favorable during gRamete implantation into the uterine mucosa. The same study demonstrates that anandamide levels are high during weeks 4-5 of gestation and decrease to lower levels during the sixth week of pregnancy.
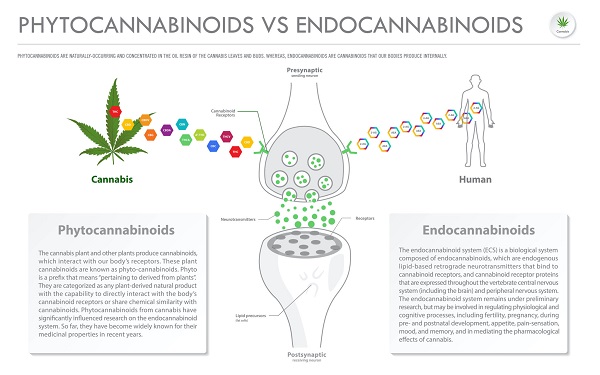
Phytocannabinoids vs Endocannabinoids horizontal business infographic illustration about cannabis as herbal alternative medicine and chemical therapy, healthcare and medical science vector.
The concentration of anandamide in the oocytes strongly correlates with oocyte maturation and quality, which determines the success of ovulation. There is currently no information on what mechanism anandamide controls oocyte maturation; the fact that high levels of anandamide are now required seems to contradict the conclusion that THC use can impair oogenesis and ovulation.
The above process may be disrupted by the role of the endocannabinoid system in digestion. Cannabinoids are known to affect key physiological processes such as appetite and glucose metabolism, thereby potentially reducing the likelihood of obesity.
Obesity is closely linked to reproductive health. Thus, the positive effects of THC on digestive processes may reduce the negative effects on ovulation itself, ultimately leading to an overall positive effect on fertility.
Conclusion
Sometimes it seems that heavy cannabis use can have negative effects on human fertility and reproduction, extensive research on the subject has paved the way for a much deeper understanding of human reproductive physiology. This has opened the door to the possibility of targeted endocannabinoid-based treatments for the 10-15% of couples worldwide who suffer from poor reproductive health.
Although cannabis use can sometimes be detrimental to an individual’s reproductive capacity, on the other hand, THC can be useful as a non-invasive means of birth control in populations with fertility rates well above the norm (the rate at which new babies must be born to replace dead individuals in order to maintain overall population size).
In sum, to get a clear picture of the extent to which cannabis use actually affects overall fertility in terms of the number of children per fertile woman, we need to look at the number of children born to cannabis users compared to those who do not use. However, various other factors can influence the number of children born, including education level, socioeconomic status, and general desire for offspring.